Cracking Hydrocarbons
It
is when big hydrocarbon molecules are broken down into smaller ones. This
is done by heating the hydrocarbon in the presence
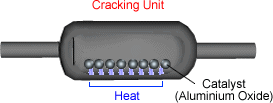
of a catalyst. This is a form of thermal decomposition. The catalyst helps to speed up the reaction. Hot vapour of hydrocarbons is passed over powdered aluminium oxide
catalyst at roughly 400ºC - 700ºC.
The long carbon chain molecules break apart or 'crack'
on the surface of the powered catalyst.
So
what is CRACKING?
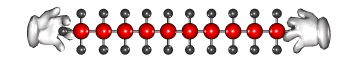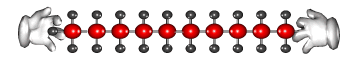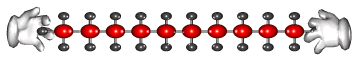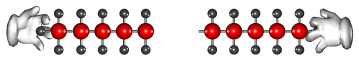
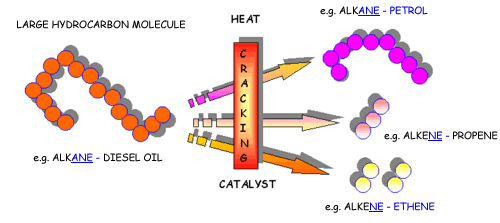
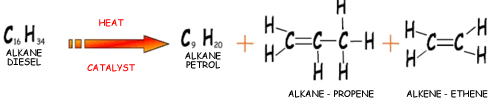
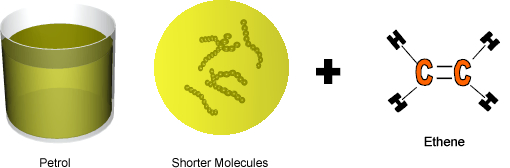
Cracking produces short-chain hydrocarbons called alkanes, like petrol and alkenes, like ethene and propene. These alkenes are very reactive due to their double bonds and therefore can be used to make other substances. Ethene, for example can be used to make ethanol and polythene.
![]()
Test your understanding of how large hydrocarbons are broken down into smaller, useful molecules. 1. What type of chemical reaction is cracking? 2. Why are large hydrocarbon molecules broken down during the cracking process? 3. Which two substances are typically produced when a large alkane is cracked? 4. What two conditions are necessary for the cracking process to take place? 5. Which of these is a common use for the alkenes produced during cracking?🧪 Knowledge Check: Cracking Hydrocarbons
Click to Reveal Answers
2. Higher demand for smaller molecules (Small molecules like petrol are more useful and in shorter supply).
3. A smaller alkane and an alkene (Cracking splits the long chain into these two types of molecules).
4. High temperature and a catalyst (These are required to break the strong chemical bonds).
5. Making plastics (Alkenes are valuable raw materials for the plastics industry).
Tags:Hydrocarbons, Cracking, Petrol, Ethane, Molecules, Diesel, Alkenes, the organic structure methane has what formula, Catalyst