Distillation
What is distillation? Simple Distillation Distilling a salt solution .
Distillation is a way of separating a liquid (the solvent) from a mixture and collecting the liquid part. Distillation involves heating the solution until it boils and then cooling the vapour until it turns back into a liquid.
For example, impure water can be distilled to produce pure water, which is safe to drink.
Distillation involves 2 stages and both are physical state changes.
(1) The liquid or solution mixture is boiled to vaporise the most volatile component in the mixture (liquid ==> gas). The ant-bumping granules give a smoother boiling action.
(2) The vapour is cooled by cold water in the condenser to condense (gas ==> liquid) it back to a liquid (the distillate) which is collected.
This can be used to purify water because the dissolved solids have a much higher boiling point and will not evaporate with the steam, BUT it is too simple a method to separate a mixture of liquids especially if the boiling points are relatively close.
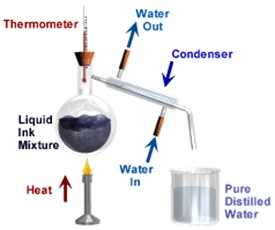
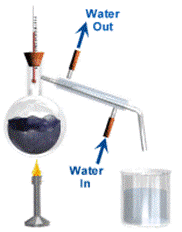
A salt solution (salt water) can be turned into pure water which we can drink. This is done by following these steps:
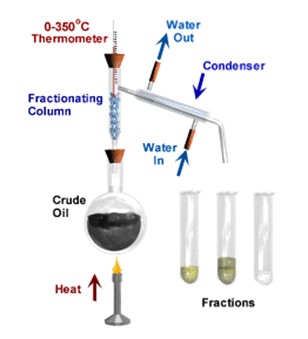
Fractional Distillation
Fractional distillation involves 2 main stages and both are physical state changes. It can only work with liquids with different boiling points. However, this method only works if all the liquids in the mixture are miscible (e.g., alcohol/water, crude oil etc.) and do NOT separate out into layers like oil/water.
(1) The liquid or solution mixture is boiled to vaporise the most volatile component in the mixture (liquid ==> gas). The ant-bumping granules give a smoother boiling action.
(2) The vapour passes up through a fractionating column, where the separation takes place (theory at the end). This column is not used in the simple distillation described above.
(3) The vapour is cooled by cold water in the condenser to condense (gas ==> liquid) it back to a liquid (the distillate) which is collected.
This can be used to separate alcohol from a fermented sugar solution.
It is used on a large scale to separate the components of crude oil, because the different hydrocarbons have different boiling and condensation points (see oil).
Test your understanding of how distillation works to separate mixtures based on boiling points. 1. In simple distillation, what is the first stage of the process? 2. Why is water passed through the outer jacket of a Liebig condenser? 3. When distilling salty water, what is left behind in the heated flask? 4. Which piece of equipment is used to monitor the temperature of the gas before it enters the condenser? 5. Simple distillation is best used to obtain: Summary: 🧪 Knowledge Check: Simple Distillation
Click to Reveal Answers
2. To cool the gas (The cold water jacket removes heat from the vapour).
3. Salt (Because salt has a much higher boiling point than water).
4. A Thermometer (To ensure the vapour is at the correct boiling point).
5. A liquid (solvent) from a solution (Such as getting pure water from sea water).