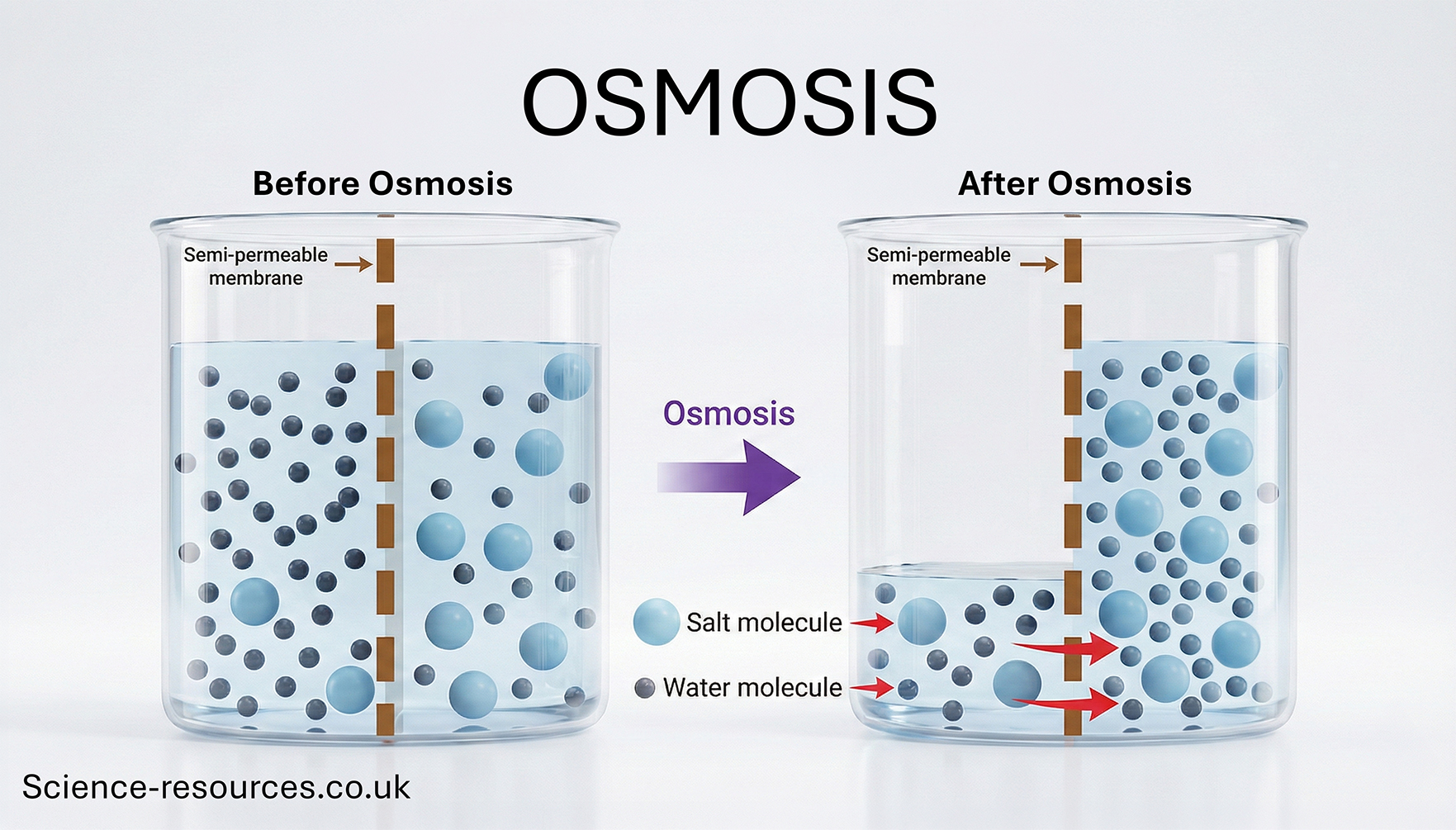
Osmosis - this is a special case of diffusion for water
Osmosis is the movement of water particles across
a semi-permeable membrane (partially permeable membrane PPM)
from an area of high water concentration to an area of low water concentration. Cell membranes are partially permeable because they will only permit some
things to pass through, but not others. The reason for this is that
PPM has extremely small pores in it. They are so small that only water
particles can pass through them, and larger particle like glucose and proteins
can not.
Experiment to demonstrate osmosis! You can demonstrate this effect with VISKING
TUBING, which is partially permeable membrane. It is
sometimes called dialysis tubing as it is used in
kidney dialysis machines. From the animation it can be seen
that water level in the glass tube begins to rise or fall because water either enters or leaves through the visking
tubing by osmosis. Remember: Potato
Slices It can be seen from the animation
that potato slices, in pure water, swell up as water
enters their cells by osmosis.
However, in concentrated
sugar solution the potato slices shrink as water
leaves their cells as a result of osmosis.
Test your understanding of how water moves in and out of cells. 1. Osmosis is the movement of which specific molecule? 2. In osmosis, water moves from a ______ solution to a ______ solution. 3. What describes a membrane that only lets small molecules pass through? 4. If a plant cell loses too much water and the cell membrane pulls away from the cell wall, the cell is: 5. Unlike active transport, osmosis is a 'passive' process. What does this mean?💧 Knowledge Check: Osmosis
Click to Reveal Answers
2. Dilute to Concentrated (Water moves to 'water down' the solute).
3. Partially permeable (Like a sieve for molecules).
4. Plasmolysed (This happens in very salty or sugary solutions).
5. Does not require energy (It follows the concentration gradient naturally).
Tags:Osmosis, Movement of water particles, Semi-permeable membrane, Cells